NCERT Solutions for Class 10 Science Chapter 4 Carbon and its Compounds are part of NCERT Solutions for Class 10 Science. Here we have given NCERT Solutions for Class 10 Science Chapter 4 Carbon and its Compounds.
| Board | CBSE |
| Textbook | NCERT |
| Class | Class 10 |
| Subject | Science |
| Chapter | Chapter 4 |
| Chapter Name | Carbon and its Compounds |
| Number of Questions Solved | 28 |
| Category | NCERT Solutions |
NCERT Solutions for Class 10 Science Chapter 4 Carbon and its Compounds
NCERT Questions
Question 1.
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Answer.
The atomic number (Z) for carbon is six and its electronic configuration is 2, 4. Carbon has four valence electrons. Each oxygen atom (Z = 8) has six valence electrons (2, 6). In order to complete its octet, the carbon atom shares its four valence electrons with the four electrons of two oxygen atoms as follows:
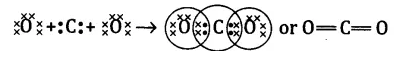
Question 2.
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur?
Answer.
The atomic number (Z) of sulphur is sixteen and its electronic configuration is 2, 8, 6. The sulphur atom has six valence electrons. The chemical formula of sulphur molecule is S8. Each sulphur atom is linked to similar atoms on either sides by single covalent bonds and thus, completes its octet. The molecule is in the form of a ring also represented by a crown shape.

You can also Download NCERT Solutions for class 10 Science in Hindi to help you to revise complete Syllabus and score more marks in your examinations.
Question 3.
How many structural isomers can you draw for pentane?
Answer.
Pentane (C5H12) has a skeleton of five carbon atoms. It can exist as a straight chain as well as two branched chains. There are three structural isomers for the hydrocarbon which is an alkane.
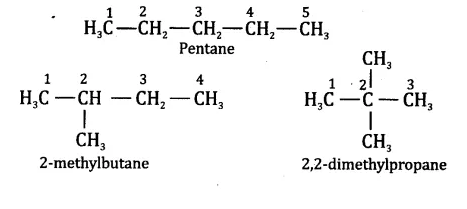
Question 4.
What are the two properties of carbon which lead to the huge number of carbon compounds we see around us? [2013]
Answer.
(a) Catenation: Carbon has the unique property of self-linking which is known as catenation. In fact, any number of carbon atoms can be linked to one another by covalent bonds. This is on account of the stability of the C—C bonds since the size of the carbon atom is quite small.
(b) Linking of carbon with other atoms: Carbon is tetravalent in nature and can readily unite with atoms such as hydrogen, oxygen, nitrogen, sulphur, etc. by electron sharing.
Question 5.
What will be the formula and electron dot structure of cyclopentane?
Answer.
Cyclopentane is a cyclic compound with the formula C5H12. The structure of the compound may be represented as:

Question 6.
Draw the structufes of the following compounds:
(a) Ethanoic acid
(b) Bromopentane
(c) Butanone
(d) Hexanal
Are structural isomers possible for bromopentane?

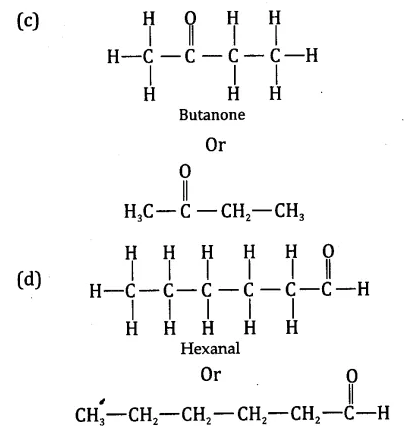
Bromopentane has a chain of five carbon atoms. It can exist in a number of forms which are structural isosmers.
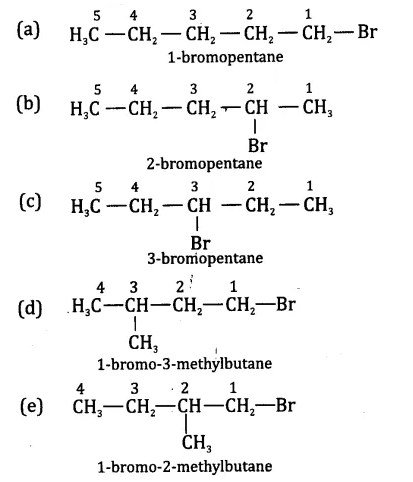

- The structural isomers (a), (b) and (c) which differ in the position of the Br atom are known as position isomers.
- The structural isomers (d), (e) and (f) which differ in the arrangement of carbon atoms in the chain are called chain isomers.
- In writing the IUPAC name, the prefix bromo is written before methyl. In fact, alphabetical order is followed while naming the different prefixes.
Question 7.
How would you name the following compounds?

Answer.
(a) Bromoethane
(b) Hex-1-yne
(c) Methanal
Question 8.
Why is the conversion of ethanol into ethanoic acid an oxidation reaction?
Answer.
Ethanoic acid (CH3COOH) has one oxygen atom more and two hydrogen atoms less than ethanol (C3H5OH). In general,
- Loss of hydrogen is known as oxidation.
- Gain of oxygen is known as oxidation.
Therefore, it is an oxidation reaction.
Question 9.
A mixture of ethyne and oxygen is used for welding. Can you tell why a mixture of ethyne and air is not used?
Answer.
When ethyne is burnt in oxygen, a large quantity of heat along with light is produced. The heat evolved can be used for gas welding which is usually carried to weld small broken pieces of articles made up of iron.
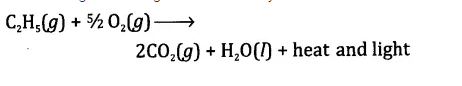
Air mainly contains of a mixture of nitrogen (4 parts) and oxygen (1 part). As we know, nitrogen gas does not support combustion. This means that in air, only oxygen will help in the combustion of ethyne. Therefore, it is always better to use oxygen for the combustion of ethyne.
Question 10.
How would you distinguish experimentally between an alcohol and a carboxylic acid?
Answer.
The distinction can be made by the following, tests:
(a) Dip a strip of blue litmus paper separately in both alcohol and carboxylic acid taken in two glass tubes. The colour will change to red in the tube containing carboxylic acid and not in the tube which contains alcohol.
(b) Add a small amount of solid sodium hydrogen carbonate (NaHCO3) in both the tubes. A brisk effervescence accompanied by bubbles will be noticed in the tube containing carboxylic acid and not in the tube containing alcohol.
Question 11.
What are oxidising agents?
Answer.
Oxidising agents are the substances which either on their own or on reacting with another substance release oxygen in order to carry oxidation reactions. The commonly used oxidising agents are: ozone, bromine water, a mixture of potassium dichromate and sulphuric acid or a mixture of potassium permanganate and sulphuric acid, etc.
Question 12.
Will you be able to check if water is hard by using a detergent?
Answer.
No, it is not possible. Detergents produce foam in any type of water; whether hard or soft Therefore, a distinction between the two cannot be made. However, soaps can be used for this purpose.
Question 13.
People use different methods to wash clothes. Usually after adding the soap, they beat the clothes on a stone or beat them with a paddle, scrub with a brush or the mixture is agitated in a washing machine. Why is this agitation necessary to get clean clothes?
Answer.
The purpose of soap or detergent in washing is to reduce friction between the oil drops carrying dirt particles and the water so that they may mix with each other. All the methods that have been suggested loosen the bonds between the dust or oil particles and fabrics of clothes. The agitation helps in washing the clothes.
Chapter End Questions
Question 1.
Ethane, with the molecular formula C2H6 has:
(a) 6 covalent bonds
(b) 7 covalent bonds
(c) 8 covalent bonds
(d) 9 covalent bonds
Answer.
(b) 7 covalent bonds
Question 2.
Butanone is a four carbon compound with the functional group
(a) carboxylic acid
(b) aldehyde
(c) ketone
(d) alcohol
Answer.
(c) ketone
Question 3.
While cooking, if the bottom of the vessel is blackened on the outside, it means that
(a) the food is not cooked completely
(b) the fuel is not burning completely
(c) the fuel is wet
(d) the fuel is burning completely.
Answer.
(b) the fuel is not burning completely
Question 4.
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Answer.
The molecule of chloromethane (CH3Cl) consists of three elements, i.e. carbon (Z = 6), hydrogen (Z = 1) and chlorine (Z = 17). Carbon atom has four valence electrons (2, 4); hydrogen has one while chlorine has seven electrons in the valence shell (2, 8, 7). In order to complete its octet, carbon shares three valence electrons with three hydrogen atoms while one is shared with the electron of chlorine atom. The structure of covalent molecule may be written as follows:

Question 5.
Draw the electron dot structures for
(a) ethanoic acid
(b) H2S
(c) propanone
(d) F2

Question 6.
What is a homologous series? Explain with an example.
Answer.
A homologous series is a series of carbon compounds that have different numbers of carbon atoms but contain the same functional group. It can be represented by the same general formula. Compounds of homologous series differ by CH2 from their consecutive members. All the compounds of a homologous series show similar chemical and physical properties. For example, alkanes such as methane, ethane, propane, butane, etc. belong to the same homologous series. Similarly, all alkenes belong to a particular homologous series and all alkynes belong to another homologous series.
Question 7.
How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties? [2011,2014]
Answer.
Distinction based on physical properties are as:
(a) Smell: Ethanol has a characteristic smell known as alcoholic smell which is pleasant. Ethanoic acid has a vinegar-like smell.
(b) Boiling point: Boiling point of ethanol (351 K) is less than that of ethanoic add (391 K).
(c) Litmus test: Ethanol is neutral in nature and does ijot change in the colour of litmus whether blue or red. Ethanoic acid is acidic and changes the colour of a blue litmus strip to red.
Distinction based on chemical properties:
(a) Action with sodium hydrogen carbonate: On adding a small amount of sodium hydrogen carbonate to ethanoic acid, carbon dioxide gas is evolved with brisk effervescence. However, no such reaction is noticed in case of ethanol.
(b) Action with caustic alkali: Ethanoic add reacts with both sodium hydroxide (NaOH) and postassium hydroxide (KOH) to form corresponding salt and water. Ethanol fails to react with either of these.
Question 8.
Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also? [2011]
Answer.
Soap may be represented by the formula RCOONa where R is an alkyl group which represents a long chain of carbon with fifteen or more atoms. Now, oil drops containing dirt particles and water do not mix. Soap helps in their mixing by reducing interfacial tension or friction. Actually it forms a sort of bridge between oil drops and water in which the alkyl portion (hydrophobic end) points towards oil drop while other portion COON a (hydrophilic end) is directed towards water. This is known as micelle formation. Thus, soap helps in the formation of a stable emulsion between oil and water. Ethanol and other similar solvents which are of organic nature do not help in micelle formation because soap is soluble in them.
Question 9.
Why are carbon and its compounds used as fuels in most cases?
Answer.
Carbon burns in oxygen to form carbon dioxide gas. The reaction is highly exothermic. That is why different forms of coal are used as fuels. The most important compounds of carbon are hydrocarbons. Just like carbon, hydrogen also readily burns in oxygen or air to form water a heat. The hydrocarbon methane (CH4) is the major constituent of natural gas. Propane (C3H8) and butane (C4H10) are present in liquid petroleum gas (LpG). Petrol and kerosene also contain different hydrocarbons. Therefore, these are used as fuels.
Question 10.
Explain the formation of scum when hard water is treated with soap. [2011,2012]
Answer.
Soap is basically sodium or potassium salts of higher fatty acids. Hard water contains Ca2+ and Mg2+ ions as their salts. When soap is added to hard water, the corresponding calcium and magnesium salts are formed. These are in the form of precipitates, also called scum.
 where R is alkyl group having 15 to 17 carbon atoms.
where R is alkyl group having 15 to 17 carbon atoms.
Question 11.
What change will you observe by testing soap with litmus paper (blue or red)?
Answer.
When soap is dissolved in water, the solution is alkaline in nature due to the formation of alkali NaOH or KOH. The solution changes the colour of red litmus to blue. However, the solution does not change the colour of blue litmus.
Question 12.
What is hydrogenation? What is its industrial application?
Answer.
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic compounds. Catalytic hydrogenation has diverse industrial uses. Most frequently, industrial hydrogenation relies on heterogeneous catalysts.
In petrochemical processes, hydrogenation is used to convert alkenes and aromatics into saturated alkanes (paraffins) and cycloalkanes (naphthenes), which are less toxic and less reactive. For example, mineral turpentine is usually hydrogenated, hydro cracking of heavy residues into diesel, etc.
Question 13.
Which of the listed hydrocarbons undergo addition reactions: C2H6, C3H8, C3H6, C2H2 and CH4?
Answer.
C3H6 and C2H2 undergo addition reaction as they are unsaturated hydrocarbons. As unsaturated hydrocarbons have double and triple bonds.
Question 14.
Give a test that can be used to differentiate between butter and cooking oil.
Answer.
Butter is saturated in nature while cooking oil is unsaturated. This means that cooking oil has at-least one C—C bond present in the constituting compounds while butter does not have any such bond. The distinction between them can be made by reacting with bromine water or bromine dissolved in carbon tetrachloride. Cooking oil will discharge the yellow colour of bromine while butter will not.
Question 15.
Explain the mechanism of cleansing action of soap.
Answer.
Cleansing Action of Soap:
Soaps contain two chemically distinct parts: a long hydrocarbon tail and the other negatively charged head. The long hydrocarbon tail is nonpolar and water repelling, i.e. insoluble in water.
When a soap is dissolved in water, the molecules clump together in a spherical fashion as clusters called micelles. The tail stick inwards and the head outwards.
In cleaning, the long hydrocarbon tail attaches itself to oily or dirty part which is nonpolar and insoluble in water. The dirt is, thus, enveloped in the nonpolar end of soap micelles. The negatively charged heads of soap molecules make the micelles soluble in water. Thus, the dirt is washed away with soap.

We hope the NCERT Solutions for Class 10 Science Chapter 4 Carbon and its Compounds help you. If you have any query regarding NCERT Solutions for Class 10 Science Chapter 4 Carbon and its Compounds, drop a comment below and we will get back to you at the earliest.