Haloalkanes And Haloarenes Class 12 NCERT Solutions includes all the important topics with detailed explanation that aims to help students to understand the concepts better. Students who are preparing for their Class 12 exams must go through NCERT Solutions for Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes. Going through the solutions provided on this page will help you to know how to approach and solve the problems.
Students can also find NCERT intext, exercises and back of chapter questions. Also working on Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes NCERT Solutions will be most helpful to the students to solve their Homeworks and Assignments on time. Students can also download Haloalkanes And Haloarenes Class 12 NCERT Solutions PDF to access them even in offline mode.
Haloalkanes And Haloarenes Class 12 NCERT Solutions
NCERT Solutions for Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes are been solved by expert teachers of CBSETuts.com. All the solutions given in this page are solved based on CBSE Syllabus and NCERT guidelines.
Haloalkanes And Haloarenes Class 12 NCERT Solutions – INTEXT Questions
Question 1.
Write the structures of the following compounds :
(i) 2-Chloro-3-methylpentane
(ii) 1-Chloro-4-ethylcydohexane
(iii) 4-terf-Butyl-3-iodoheptane
(iv) 1, 4-Dibromobut-2-ene
(v) 1 -Bromo-4-sec-butyl-2-methylbenzene
Solution:

Haloalkanes And Haloarenes Class 12 NCERT Solutions Question 2.
Why is sulphuric acid not used during the reaction of alcohols with Kl ?
Solution:
H2SO4 is a strong oxidising agent. Therefore, when it is used in presence of KI, it tends to convert KI to HI and finally oxidises it to I2.
Question 3.
Write structures of different dihalogen derivatives of propane.
Solution:
The structures of all possible dihalogen derivatives of propane are

Haloalkanes And Haloarenes Class 12 NCERT Solutions Question 4.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields
(i) A single monochloride
(ii) Three isomeric monochlorides
(iii) Four isomeric monochlorides.
Solution:
An alkane with molecular formula C5H12 can exist in the following isomeric forms :



Haloalkanes And Haloarenes Class 12 NCERT Solutions Question 5.
Draw the structures of major monohalo products in each of the following reactions :

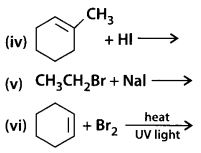
Solution:
The major haloderivatives formed in the given reactions are

Question 6.
Arrange each set of compounds in order of increasing boiling points.
(i) Bromomethane, bromoform, chloromethane, dibromomethane.
(ii) 1 – Chloropropane, isopropyl chloride, 1 – chlorobutane.
Solution:
The boiling points of organic compounds depend on the strength of the intermolecular forces in them. These forces are :
(a) van der Waals forces and
(b) dipole-dipole interactions These forces are dependent on the
(i) molecular mass and
(ii) surface area of the molecules
(i) As the molecular mass of the compound increases, the boiling point also increases. Therefore the correct order is
chloromethane < bromomethane < dibromomethane < bromoform
(ii) Amongst molecules with same mass, it is the size of the molecule that determines the boiling point. Branched compounds are more compact and therefore have less surface area as compared to their straight chain counterparts and therefore lower boiling point. The order of boiling point is
iso-propyl chloride < 1-chloropropane < 1-chlorobutane
Question 7.
Which alkyl halide from the following pairs would you expect to react more rapidly by an SN2 mechanism? Explain your answer.

Haloalkanes And Haloarenes Class 12 NCERT Solutions:
We know that SN2 mechanism involves a transition state wherein both, the incoming nucleophile as well as the leaving group are present around the carbon atom. There are 5 atoms simultaneously bonded to it.
Thus, for such a transition state to be possible, there should be minimum steric hindrance. Hence, 1° alkyl halides are most reactive towards SN2 followed by 2° and finally 3°.
1° RX > 2° RX > 3° RX

![]()
Question 8.
In the following pairs of halogen compounds, which compound undergoes faster SN1 reaction?

Haloalkanes And Haloarenes Class 12 NCERT Solutions:
SN1 reaction proceeds via the formation of a carbocation intermediate. This intermediate is formed by the cleavage of the C — X bond. More stable is the resultant carbocation faster is the SN1 reaction.
Order of stability of carbocation is
3° carbocation > 2° carbocation >1° carbocation

Question 9.
Identify A, B, C, D, E, R and R’ in the following.

Solution:



10. A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Ans. The hydrocarbon with molecular formula C5H10 can either a cycloalkane or an alkene.Since the compound does not react with Cl2 in the dark, therefore it cannot be an alkene but must be a cycloalkane. Since the cycloalkane reacts with Cl2 in the presence of bright sunlight to give a single monochloro compound, C5H9Cl, therefore, all the ten hydrogen atoms of the cycloalkanes must be equivalent. Thus, the cycloalkane is cyclopentane.

NCERT Exercises
Question 1.
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides :
(i) (CH3)2CHCH(Cl)CH3
(ii) CH3CH2CH (CH3)CH(C2H5)Cl
(iii) CH3CH2C(CH3)2CH2l
(iv) (CH3)3CCH2CH(Br)C6H5
(v) CH3CH(CH3)CH(Br)CH3
(vi) CH3C(C2H5)2CH2Br
(vii) CH3C(CI)(C2H5)CH2CH3
(viii) CH3CH = C(Cl)CH2CH(CH3)2
(ix) CH3CH = CHC(Br)(CH3)2
(x) p-ClC6H4CH2CH(CH3)2
(xi) m-CICH2C6H4CH2C(CH3)3
(xii) o-BrC6H4CH(CH3)CH2CH3
Haloalkanes And Haloarenes Class 12 NCERT Solutions:



Question 2.
Give the IUPAC names of the following compounds:
(i) CH3CH(Cl)CH(Br) (CH3)
(ii) CHF2CBrClF
(iii) ClCH2C = CCH2Br
(iv) (CCl3)3CCl
(v) CH3C(p-ClC6H4)2CH(Br)CH3
(vi) (CH3)3CCH = C(CI) C6H4 l-p
Solution:


Question 3.
Write the structures of the following organic halogen compounds.
(i) 2-Chloro-3-methylpentane
(ii) p-Bromochlorobenzene
(iii) 1 -Chloro-4-ethylcyclohexane
(iv) 2-(2-Chlorophenyl)-1 -iodooctane
(v) Perfluorobenzene
(vi) 4-tert-Butyl-3-iodoheptane
(vii) 1 -Bromo-4-sec-butyl-2-methylbenzene
(viii) 1, 4-Dibromobut-2-ene
Solution:
Structures of the given compounds are :


Question 4.
Which one of the following has the highest dipole moment?
(i) CH2Cl2
(ii) CHCl3
(iii) CCl4
Solution:


Question 5.
A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Solution:
A number of structural isomers are possible for molecular formula C5H10. But, the given compound gives a single monochloro derivative when reacted with Cl2 in sunlight suggests that, all the H-atoms in the compound are equivalent. This is possible only if the compound is a cyclic alkane.

Question 6.
Write the isomers of the compound having formula C4H9Br.
Solution:
The possible isomers of C4H9Br are

Question 7.
Write the equations for the preparation of 1 -iodobutane from
(i) 1-butanol
(ii) 1-chlorobutane
(iii) but-1-ene
Solution:

Question 8.
What are ambident nucleophiles? Explain with an example.
Solution:
Ambident nucleophiles are nucleophiles that are capable of attacking the substrate (alkyl halide) through two different atoms.
It so ‘happens due to the presence of two nucleophilic centres which arise from the contributing (resonance) structures that are possible for the ion.
e.g., In NO–2 ion, there is a lone pair of electrons on N and therefore makes it nucleophilic while oxygen by virtue of the negative charge acts as a nucleophile. Thus, NO–2 can attack via O or N atom thereby making it ambidentate.

Question 9.
Which compound in each of the following pairs will react faster in SN2 reaction with OH– ?
(i) CH3Br or CH3l
(ii) (CH3)3CCI or CH3Cl
Solution:
(i) Between CH3Br and CH3I, CH3I will react faster via the SN2 mechanism. In SN2 mechanism, C – X bond breaks and the faster it breaks faster is the reaction.
I- is a better leaving group. Owing to its large size, the C – I bond breaks faster than the C – Br bond and reaction proceeds further at a greater rate.
(ii) The order of reactivity in an SN2 reaction depends on minimal steric hindrance around the carbon involved in the C – X bond. Lesser the steric hindrance felt by the incoming nucleophile, more reactive will be the alkyl halide towards SN2 reaction.
Based on this, CH3Cl will react faster than (CH3)3CCl.
Question 10.
predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene :
(i) 1-Bromo-1-methylcyclohexane
(ii) 2-Chloro-2-methylbutane
(iii) 2, 2, 3-Trimethyl-3-bromopentane.
Solution:


Question 11.
How will you bring about the following conversions?
- Ethanol to but-1 -yne
- Ethane to bromoethene
- Propeneto 1-nitropropane
- Toluene to benzyl alcohol
- Propene to propyne
- Ethanol to ethyl fluoride
- Bromomethane to propanone
- But-1 -ene to but-2-ene
- 1-Chlorobutane ton-octane
- Benzene to biphenyl
Solution:




Question 12.
Explain why
- the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?
- alkyl halides, though polar, are immiscible with water?
- Grignard reagents should be prepared under anhydrous conditions?
Solution:
(i) (a) In order to understand the lower dipole moment of chlorobenzene we need to look into the contributing structures of the molecules.

(b) From the above structures we find that the C – Cl bond in chlorobenzene has a partial double bond character (structure II, III and IV). As a result, the C – Cl bond length here is shorter than the C – Cl single bond but longer than the C – Cl double bond.
(c) Also evident is the positive charge on Cl atom which reduces the partial negative (δ-) charge which it is expected to carry by the virtue of its electronegativity.
(d) Consequently, the dipole moment, which is a product of bond length and partial negative charge on Cl atom, reduces. However, in cyclohexyl chloride this does not happen. It is an alkyl halide and carbon is purely sp3 hybridised and C – Cl bond has the bond length of a single bond and 8“ appearing on Cl is also higher, thus, the greater dipole moment.
(ii) Only those compounds which can form hydrogen bonds with water are miscible with it. Alkyl halides, though polar due to the presence of electronegative halogen atom, are immiscible since they cannot form hydrogen bonds.
(iii) Grignard reagents R – Mg – X is a class of highly reactive compounds which can extract a proton even from water molecule. They thus, turn into the corresponding alkanes and render any other desired reaction ineffective.
This is why Grignard reagents are prepared in the absolute absence of water (anhydrous conditions), (e.g.,)

Question 13.
Give the uses of freon 12, DDT, carbon tetrachloride and iodoform.
Solution:
(i) Freon 12 (CCl2F2) is
- used in aerosol propellants
- refrigeration
- air-conditioning.
(ii) DDT (p, p’- dichlorodiphenyltrichloroethane) is
- used as an insecticide,
- mainly used against mosquitoes.
(iii) Carbontetrachloride (CCl4) is used
- in manufacture of refrigerants and propellants for aerosol cans
- in synthesis of chlorofluorocarbons
- as degreasing agent
- as cleansing agent
- as a solvent in laboratories
(iv) Iodoform (CHI3) is used as an antiseptic.
Question 14.
Write the structure of the major organic product in each of the following reactions :

Solution:



Question 15.
Write the mechanism of the following reaction
![]()
Solution:

Question 16.
Arrange the compounds of each set in order of reactivity towards SN2 displacement :
- 2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
- 1-Bromo-3-methylbutane, 2-Bromo-2- methylbutane, 3-Bromo-2-methylbutane
- 1-Bromobutane, 1-Bromo-2,2-dimethylpropane, 1-Bromo-2-methyl butane, 1-Bromo-3- methylbutane.
Solution:
SN2 reaction proceeds via the formation of transition state where the carbon atom is surrounded by 5 other atoms (groups). Thus, for such a transition state to form, the steric interactions have to be minimum. Therefore, the most favourable substrates for SN2 reactions are 1° alkyl halides followed by 2° and 3° alkyl halide. Order of reactivity towards SN2 : 1° > 2° > 3° > aryl halide.


In example (iii), although all the given alkyl halides are 1° but the steric hindrance around the carbon bearing the -Br atom decides the order of reactivity. More the number of bulky groups around this carbon lower will be its reactivity towards SN2.
Question 17.
Out of C6 H5 CH2 Cl and C6H5 CHClC6 H5, which is more easily hydrolysed by aqueous KOH?
Solution:
C6 H5 CHClC6 H5 is hydrolysed faster.
(a) Hydrolysis of an alkyl halide is an example of nucleophilic substitution reaction. In case of aryl halides this follows the SN1 pathway i.e., via the formation of carbocation.
(b) C6 H5 CH2 Cl or benzyl chloride gives

(c) Out of I & II, carbocation II is more stable. The reason is the presence of two phenyl rings attached to the carbon carrying the positive charge.
(d) As a result, the delocalisation of the +ve charge is greater and the carbocation is more stable. Due to this, (II) is formed faster and the corresponding halide is hydrolysed with greater ease as compared to benzyl chloride.
Question 18.
p-Dichlorobenzene has higher m.p. and solubility than those of o- and m-isomers. Discuss.
Solution:
The para-isomers have high melting points and solubility as compared to their ortho and meta isomers due to symmetry of para-isomers that fits into crystal lattice better than ortho and para isomers.
Question 19.
How can the following conversions be carried out?
- Propene to propan-1 -ol
- Ethanol to but-1 -yne
- 1 -Bromopropane to 2-bromopropane
- Toluene to benzyl alcohol
- Benzene to 4-bromonitrobenzene
- Benzyl alcohol to 2-phenylethanoic acid
- Ethanol to propanenitrile
- Aniline to chlorobenzene
- 2-Chlorobutane to 3,4-dimethylhexane
- 2-Methyl-1 -propene to 2-chloro-2- methylpropane
- Ethyl chloride to propanoic acid
- But-1-ene to n-butyliodide
- 2-Chloropropaneto 1-propanol
- Isopropyl alcohol to iodoform
- Chlorobenzene to p-nitrophenol
- 2-Bromopropane to 1-bromopropane
- Chloroethane to butane
- Benzene to diphenyl
- ferf-Butyl bromide to isobutyl bromide
- Aniline to phenylisocyanide
Solution:







Question 20.
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Solution:
Formation of alcohols from the reaction between alkyl chlorides and aqueous KOH is an example of simple nucleophilic substitution.

But when aqueous KOH is replaced by alcoholic KOH, alkenes are formed instead of alcohols due to elimination of HCl from an alkyl halide.
![]()


Question 21.
Primary alkyl halide C4H9Br (A) reacted with alcoholic KOH to give compound (B). Compound (B) is reacted with HBr to give (C) which is an isomer of (A). When (A) is reacted with sodium metal it gives compound (D). C8H18 which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (A) and write the equations for all the reactions.
Solution:


Question 22.
What happens when
- n-butyl chloride is treated with alcoholic KOH,
- bromobenzene is treated with Mg in the presence of dry ether,
- chlorobenzene is subjected to hydrolysis,
- ethyl chloride is treated with aqueous KOH,
- methyl bromide is treated with sodium in the presence of dry ether,
- methyl chloride is treated with KCN?
Solution:


Chlorobenzene is highly unreactive towards nucleophilic substitution. However, it can be hydrolysed to phenol by heating in aqueous sodium hydroxide solution at a temperature of 623 K and 300 atm pressure. The presence of an electron withdrawing group increases the reactivity of haloarenes.

Now that you are provided all the necessary information regarding NCERT Solutions for Class 12 Chemistry Chapter 10 Haloalkanes and Haloarenes and we hope this detailed NCERT Solutions are helpful.