NEET Biology Notes Biomolecules and Enzyme Carbohydrates
Carbohydrates
- Carbohydrates are the basic component of food and principal source of energy, which are composed of carbon (C), hydrogen (H) and oxygen (O) in the approximate ratio of 1:2:1.
- If carbohydrates are composed of only single sugar molecule, it is called a monosaccharide (i.e. single sugar), if two or more monosaccharides are linked, they form a oligosaccharide (i.e. 2-10 units of monosaccharides) or a polysaccharide
(i.e. many monosaccharides). - Polysaccharides are long chains of sugars. They are threads containing different monosaccharides adjust as building blocks.
- For example, cellulose is a polymeric polysaccharide consisting of only one type of monosaccharide, i.e. glucose.


D-glyceraldehyde
It is three carbon, aldotriose (aldose sugar + triose sugar). It is also the smallest carbohydrate. D-Ribose is an important sugar used in genetic material. This sugar is not used as an energy source but is a part of the backbone of RNA. When OH group of C-2 position is removed (H in place of OH) from ribose the sugar becomes deoxyribose, which is used in the backbone of DNA. Pentose sugar ribose is found in every animal cell. Ribose is the main constituent of ATP, ADP, riboflavin and RNA.
Polysaccharides
Polysaccharides are long chains of monosaccharide units they may number from several into the hundreds or even thousands. The major polysaccharides of interest in nutrition are glycogen, found in certain animal tissues and starch and cellulose, both of plant origin. All these polysaccharides consist of only glucose units.
Polysaccharides may be either homopolysaccharides (i.e. containing a single type of monomers, e.g. starch, glycogen, cellulose, chitin) or heteropolysaccharides (i.e. containing two or more different units), e.g. hemicellulose, pectic substances, some gums, etc. In a polysaccharide chain, the right end is called the reducing end and the left end is called the non-reducing end.
Functions of Polysaccharides
- Glucose is the most important sugar in our diet, which is used as immediate source of energy. It is also called dextrose.
- Glucose is stored as glycogen in liver and muscles. Level in blood can be as high as 0.1 %.
- D-fructose (levulose) is a ketohexose- and sweetest of all sugars.
- Erythrose is raw material for synthesis of anthocyanin and lignin.
- Galactose in milk is a component of milk sugar lactose.
- Galactosides are compounds of galactose, occur in brain and nervous tissue.
- Galactose is a constituent of agar-agar.
Reducing Sugar
The term ‘reducing’ reflects the fact that some sugars have carbonyl groups (C.=0), which can be oxidised to carboxylic acids (—COOH), reducing other chemicals in the process.
A standard test for a reducing sugar is Benedict’s solution, a blue solution that contains copper sulphate, f a reducing sugar is present, the Cu (II) ions in copper sulphate are reduced to Cu (I) ions resulting in an orange precipitate.
Glucose, fructose, galactose, maltose and lactose are all reducing sugars but sucrose is a non-reducing sugar. However, after sucrose is boiled with dilute acid to hydrolyse (split) it into its monosaccharides, it produces a positive result.


Different Types of Sugar
(i) Sucrose or table sugar It is found in sugarcane and sugarbeet upto 20 % by mass.

Maltose or malt sugar It is not common in nature except in germinating starchy seeds.
It is referred to as p-maltose because the unreacted C-l on (3-D glucose is in the p position.
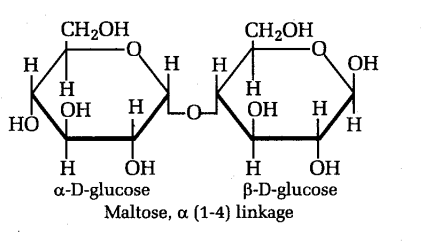
Maltose is produced commercially from starch by a starch hydrolysing enzyme diastase.
(iii) Lactose or milk sugar It is dimer of p-D galactose and either the a or p-D-glucose.

- Glycosidic Bond
The carbon that carries the aldehyde or the ketone can react with any hydroxyl group on a second sugar molecule to form a bond called glycosidic bond.
Based on the position of the C-l OH (i.e. hydroxyl (OH) group present on carbon number-1), glycosidic bonds may be
- a-glycosidic bond linkage between a C-l a OH and a C-4 OH.
- p-glycosidic bond linkage between a C-l p OH and a C-4 OH.

C-4 end can be either up or down depending on the orientation of the monosaccharide. Cellobiose consists of two molecules of p-D glucose. It cannot be split by mammalian enzymes. Cellobiose is similar to maltose except presence of p (1-4) linkage in cellobiose instead of a (1-4) in maltose. Due to presence of p (1-4) linkage, cellobiose is undigestible.
- Complex Carbohydrates
There are more complex carbohydrates in nature, e.g. exoskeleton of arthropods, have a complex polysaccharide called chitin.
Starch forms helical secondary structures. Infact, starch can hold I2 (iodine) molecules in the helical portion, the starch I2 (iodine) is blue in colour. Plant cell walls are made of cellulose. Paper made from plant pulp is cellulose. Cotton fibre is cellulose.
Some popular complex carbohydrates are :
- Glucans are polymers of glucose monomers, e.g. Starch, glycogen, cellulose, chitin. .
- Galactans are polymers of galactose monomers, e.g. Agar-agar, pectin, galacton from snails.
- Mannans are polymers of mannose monomers, e.g. yeast mannan.
- Xylans are polymers of xylose monomers, e.g. hemicellulose xylan.
- Fructans are polymers of fructose of monomers, e.g. inulin.
- Agar-agar is a galactan consisting of both D-and- L-galactose. It is used as a mircrobial culture medium.
- Pectin contains arabinose, galactose and galacturonic acid. Pectins are abundant in fruits such as orange, lemon etc.
- Pectic acid is a homopolymer of the methyl ester of D-galacturonic acid.
- Glycogen (animal starch) is commonly found in fungi and animals. It is water soluble, which gives a red colour with iodine. A starving man first consumes reserve glycogen. Almost pure cellulose is present in cotton.
- Human cannot digest cellulose. It is digested by termites and sheeps by harbouring bacteria and protozoans that synthesize the necessary enzyme cellulase.
- Inulin (Dahlia starch) is a polymer of fruclorc uuib: linked by (3-1, 2 glycosidic linkages.
- Hyalouronic acid found in skin, viterous humour of the eye, umbilical cord as a coating around the ovum and in certain bacteria are mucopolysaccharides.
- Chondriotin sulphates, predominant in cornea, cartilage, tendons, skin, heart valves and saliva are also mucopolysaccharides.
